Effect of Hyamine-1622 Cationic Surfactant on Pertraction of Cerium (IV) Cations Through Emulsion Liquid Membranes
Abstract
The present study aims to shed light directly towards the extraction of (IV) cerium ions using "liquid surfactant membrane" technology, "LSM" developed in the presence of synergistic cationic and nonionic materials. The effect of various factors such as Ce (IV) transport, synergistic surfactants, curing ratio, stir speed, temperature, and mixing time between the carrier and the cerium ion on the extraction rate was studied by LSM taking into account surfactant agents. The positive effect of benzethonium chloride "Hy-1622" on the extraction of cerium ion was demonstrated by LSMs technique. Experiments confirmed the efficiency of Hy-1622 chloride synergistically with Span 80/85 to extract cerium ions with LSMs technology for emulsions in the oil phase is critical as it determines the stability, viscosity and mass transfer resistance of the resulting emulsion. Besides, Hy-1622 chloride was found as a new cationic surfactant that appeared in FTIR characterization and surfactant was found to speed up the permeability process and accelerate the extraction rate due to electrostatic interaction with the carrier.
Author Contributions
Academic Editor: Mohammad Tavakkoli, Shahid Chamran University of Ahvaz, Ahvaz, Iran.
Checked for plagiarism: Yes
Review by: Single-blind
Copyright © 2021 Amany T. Kassem, et al.
 This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Competing interests
The authors have declared that no competing interests exist.
Citation:
Introduction
The increased discharge of various metals into the environment by different industries is a threat to the environment. Thus, recovering it from wastewater is an important topic of research in wastewater treatment. Lanthanides are among the most toxic minerals. Cerium is one of the rare chemical elements that many daily industries depend on, such as color televisions, fluorescent lamps, glasses and energy-saving lamps, and its salts can be used to stimulate the metabolism process 1, 2, 3. The danger of cerium most often lies in the workplace, because moisture and gases can be inhaled with the air. This leads to lung obstruction, especially during long-term exposure 4. Cerium can also pose a serious threat to the liver when it accumulates in the human body. Cerium is expelled into the environment, mainly by industries that produce gasoline, and when disposing of household equipment5, 6. Cerium also accumulates gradually in soil and water, which eventually leads to increased concentrations in humans, animals, and soil particles 7, 8, 9. Moreover, it damages the cell membranes of aquatic animals, leading to many harmful effects on reproduction and nervous system functions. This paper investigated the possibility of applying the removal of trivalent cerium from sewage water under acidic conditions with a cationic surfactant. Several methods are used to remove cerium from industrial wastewater which is concentration reduction followed by chemical precipitation; Ion exchange; Adsorption; Electrochemical deposition of solvent extraction; Membrane separation fixation, evaporation, reverse osmosis, foam separation, freeze separation, and bio-sorption 10, 11. This view proves that liquid surfactant membranes (LSMs) or emulsion liquid membranes (ELMs) are an effective and inexpensive method that can be applied to this goal. It was observed that this technique combines the extraction and stripping steps in one stage, due to the simultaneous purification and concentration of the solute. The effect to be addressed at the external phase is the design in contact with an emulsion dispersed into globules. Each globuleconsists of droplets from an internal aqueous receiving phase with a medium diameter of saturation encapsulated in an organic membrane phase (oil phase) containing a surfactant. The emulsion was prepared in advance by stirring at a high mixing speed (or emulsification phase). During mixing between wastewater and emulsion globules (or permeability), the solutes materials are transported across the membrane from the outer phase into the inner drops where they are concentrated 12, 13. The liquid membrane consists of a diluent, a surfactant to stabilize the emulsion, and a carrier if the solutes are separated by a chemical reaction. Consequently, the effluent from the emulsion was separated by gravity, and the demulsification was also separated 14.The emulsification was explicit, to separate the phases that make the emulsion, the internal aqueous phase, and the organic membrane phase. At the end of the technique, the membrane phase can be restored and the enriched in the recovered solute can be recycled or recovered for the solute. Due to the high surface area of the emulsion globules and internal droplets, high mass transfer rates using the LSM process are very suitable. After that, the permeability of the liquid membrane is usually accompanied by the appearance of some imperfect things such as swelling and emulsion breaking 15. Such harmful phenomena should be avoided by choosing the appropriate conditions. Several studies have been performed to separate Ce(IV) ion species by LSMs. The emulsion liquid membrane was inspected for the adoption of optimal conditions for the extraction of Ce(III) ions from acidic chloride solutions using Cyanex 301 as the extractant. The effect of various factors such as extractant, and surfactant concentrations, mixing speed, concentration, type of separation solution, phase ratio, treatment ratio, and nature of diluent, on the extraction rate was studied. Under optimal conditions, the extraction of Ce(III) ions was also studied for different feed mixture solutions. Solvent extraction and stripping of Ce(III) ions were performed to study the effect of various factors such as the pH of the aqueous solution, the concentration of Cyanex 301 16, 17, and the equilibrium time on both the extraction ratio of Ce(III) ions as well as the separation ratio of metal ions from loading organic phase by hydrochloric acid solution 18, 19. In this work, Ce(IV) extraction was performed as in nitric acid concentration, using Hy-1622 as an LSM cationic surfactant with diethyl (2-ethylhexyl) 2-ethylhexyl phosphate. The effect of membrane material, carrier concentration, and various conditions, etc. on extraction efficiency was studied. Moreover, the emulsification of oil and water (W/O) has been studied in the high electric current field. To achieve a synergistic liquid surfactant membrane process for recovery of cerium where the countercurrent process was demonstrated under the optimum conditions obtained in batch operation, and selective separation of Ce (IV) in the presence of different ions 19.
Experimental
Reagents and Methods
2-DEHEHP, B (GR grade, Merck), Benzene (BDH), 1,2-dichloroethane (ClCH2CH2Cl) (ɛ=10.50), D=17.34, dichloromethane (5% v/v) (all from Merck) Ce(IV) (GR grade. Merck), different metal ions such as Mg(II), Ca(II) and K (I) 1x10-3 M in 3 M HNO3 (GR grade. Merck), sodium hydroxide, and sodium nitrate (BDH) were used without purification. 1,2-dichloroethane, dichloromethane, and Benzethonium chloride Wt= 448.1 g/mol (all from Merck) with the highest purity were used as liquid membranes. All aqueous solutions were prepared using doubled distilled water (DDW), 20, 21. SPAN 80 and SPAN 85 with benzothonium chloride were used as surfactants in this study. HLB for cationic with non –ionic surfactants was purchased from Sigma Aldrich. Because of the lower HLB value, 5.6 for non –ionic and 13.8 for cationic are expected to stabilize W/O emulsions. Since benzothonium chloride is considered HLB 12 into 14 for this component , so the amphiphilic individuality and to its high solubility in water , the HLB of benzothonium chloride or Hy-1622 is assigned a value of more than HLB for non –ionic surfactant due to their higher HLB value are assumed to stabilize O/W emulsions.
Preparation of Emulsion
To prepare a stable emulsion membrane, the droplet size should be very small, in the range of 2.8-4 μm. This can be accomplished by image capture through high energy density into a W / O (dispersed / continuous) phase emulsion during emulsification. For the small liquid surfactant membrane system, high-speed agitators are used with stirring speeds of 4.000 rpm in the emulsion prepare. During emulsification, an internal aqueous phase containing a sodium nitrate as stripping agent is introduced. such as the extraction method with an extractant in the membrane phase, the selected sodium nitrate as stripping agent should be also thermodynamically appropriate for the stripping process and display, a fast reaction with aH+[NO3]-2 -2DEHEHP, B complex. However, due to the very large interfacial area of internal droplets as long as, even the sodium nitrate as stripping procedure, with very low reaction kinetics can be utilize in an LSM system. From Table 1 the emulsion liquid membrane (ELM) by emulsifying in the wastewater of acid strip sodium nitrate with membrane phase (Oily phase). Take 100 g of emulsion (a known amount of hydrophilic cerium-oxide particles, with extractant (2DEHEHP, B) as carrier was placed in glass beaker 250 ml. The final weight of the emulsion 1:1 (O/W). Then the pH of the solution was adjusted by using (1 M NaOH). Finally the solution completed by adding distilled water adjusted at pH required the concentration of cerium oxide particles (0.0005 M) at pH (2.5)
Table 1. Experimental conditions for Ce(IV) extraction with emulsion liquid membrane ELMs| Membrane phase(oily phase) | |
|---|---|
| Volume | 50 Cm3 |
| Carrier 2(DEHEHP) | 1x10-3M |
| Diluent (dichloromethane) | 5% v/v |
| Non-anionic surfactant | |
| Span 80 | 100 g/L |
| Span 85 | 130 g/L |
| Xylene | 50% |
| Kow = KD = X0/Xw | 3.5 proportion remains in the organic phase |
| Cationic surfactant | |
| (Hy-1622) | solubility of 160 g/L in water |
| De-ionized water | 85 ml |
| Aqueous phase | |
| Volume | 15 ml |
| Ce(IV) | 25 ml |
| pH | 1-2.5 |
| Buffer{NaNO3 HNO3} | 1X10-3 mol/L |
| Feed phase | |
| Volume | 15 ml |
| Acidity | 3M of Ce(NO3)2 |
| Feed composition | Ce(IV) with different metals such as Potassium, Magnesium &Calcium |
| Concentration | 5x10-4 mol/L 1x10-3 mol/L |
| Stirring speed | 2000-4000 rpm |
Procedure
The transport experiments utilized standardized concentric cells in which the aqueous phase (15 cm3) and receiving phase (25 cm3) were separated by an oily phase (50cm3). All experiments were carried out at 250C. The oily layer was stirred by a Teflon-coated magnetic bar at 200-400 rpm. Under better experimental conditions, the mixing process should be ideal to avoid the interfaces between the organic membrane and the two aqueous phases. The aqueous phase (AP) contained cerium (IV) nitrate (5x10-4M). The membrane phase (MP) contained the benzethonium chloride surfactant (1x10-3M) and the receiving phase (RP), consisted of sodium nitrate as a stripping agent (1x10-3 M). Through the implementation of the transport experiment, samples of aqueous phases jointly were analyzed for metal by atomic absorption spectroscopy (AAS) Model Thermo Scientific, USA), under optimal conditions λCe at 470-485 nm 21. From the experimental volume ratio for O/S (1:1) was equilibrated in the shaker after separation using a centrifuge type (Z-230) (Hermle, Germany) at 250C for 20 hr. After reaching equilibrium separated from the organic phase and raffinate sodium hydroxide, the metal loaded O/S (1:1) was equilibrated, determine the equilibrium distribution ratio for stripping into organic phases. For the emulsion system, the external phase/internal phase (W/O) was prepared and stirring speed 2000-4000rpm. Figure 1 Schematic diagram representation of the emulsification system, and Figure 2 molecular structures of 2 (DEHEHP, B), Span 80, 85 (a non-ionic surfactant) and benzethonium chloride (Hy-1622) cationic surfactant and Table 2: physical and chemical properties of non-ionic and cationic surfactants.
Figure 1.Schematic diagram representation of the emulsification system.
Figure 2.Molecular structures of (a): 2(DEHEHP,B), (b & c):Span 80,85 (non-ionic surfactant) and (d): Benzathonium chloride (Hy-1622) cationic surfactant.
| Parameter | Span 80 | Span 85 | Benzathonium chloride(Hy-1622) |
| M.Wt | 428.6 | 448.1 | |
| Type | Non-ionic | 957.52 | Cationic |
| Density | 0.986 g/ml | Non-ionic | 0.998 g/ml |
| Viscosity | Viscous liquid | 0.94 g/ml | When shaken form viscous solution |
| Sp.G | 0.986 | 200-300 mpa.s | 0.98 |
| Solubility | Soluble in fatty oils | 0.956 | Soluble in H2O at 18o C |
| Color | Yellow | Soluble in CHCl3 | White solid |
| HLB | 4.3 | Light yellow | ---- |
| Stability | Stable, incomplete with strong oxidizing agents | 1.8±1.1 | Stable but hygroscopic, ncompatible with strong oxidizing agents |
Emulsion Stability Study
Emulsion stability plays an important role in the implementation of emulsion surfactant membrane ESM, it is decisively to culminate the desired level of stability. In this work, the optimum conditions for stable emulsion were determined practically. The stability study of the emulsion was carried out by manipulating different parameters like stripping rate, mixing stirring, treat ratio Vm/Vce , the effect of temperatures, and the effect of viscosity. The viscosities of the chemicals and mixtures were measured with a Vibro viscometer Brookfield RVDVE230 Medium-range viscometer), all viscosity measurements were done in thermostated conditions, at 45oC, except when the temperature influence was studied. Viscosity measurement systems used corresponding to the three apparatuses and the different conditions sets of rotation speed and spindle number. In each case, the dynamic viscosity/Absolute viscosity below 1. All results are shown in Table 4. At various temperatures and the density measuring by using the Holman method. The external feed phase at a carrier initial concentration was mixed with prepared surfactant and stirred. Across accomplishment of agitation, the fraction of the external phase at pH 2.5. Membrane breakage, ε (%) were calculated based on the change in the pH more than 1.8 or less than 3 according to equations 1 in the external phase, can be determined by pH meter according to the following equation
 ...(1)
...(1)
Where ƹ% membrane breakage, Vi initial volume of the internal phase,Ve volume of the external phase.
Measuring of Surfactant
Most of the types of surfactant added during emulsification adsorb to connect, between the membrane phase and the stripping phase. Therefore, an increase in the surfactant concentration in the membrane phase enhances the strength of the adsorption layer. When surfactant concentration increases due to a decrease in the interfacial tension between the membrane phase and the stripping phase 19, 20. The interfacial tension is intermolecular interactions, which exist between two immiscible liquid phases such as oil and water. The energy barrier produced by the interfacial tension prevents forming an emulsion. Hence, the interfacial tension should be lowered by adding an emulsifier (e.g. surfactant) for the emulsification. Because of the lowered interfacial tension by surfactant, the size of the internal droplets can be decreased, which consequently enhances the emulsion stability. There is a critical surfactant concentration beyond which the emulsion stability is improved no more. However, this remedy is associated with a couple of critical problems in the LSM process. Firstly, the standard of high surfactant causes swelling of emulsion, which is explained by one of the swelling mechanisms mentioned above: entrainment of the external feed phase by immoderate surfactant in the oily phase and by transferred micelles of surfactant. Also, high surfactant levels can hinder the mass transfer of the solute by increasing the interfacial resistance and interfere with the carrier reaction at the interface.
Result and Discussion
Characterization of Hyamine-1622
The new cationic surfactant, Hy-1622 and characterized in the solid-state by FT-IR, and in solution by1H-and13C-NMR spectroscopy. The value of (CMC) or critical micelle concentration was determined by UV-visible spectroscopy. Interaction of surfactant with cerium ion was studied by UV-visible spectroscopy. These surfactants were proved to be efficient in increasing the solubility of cerium molecules. To test the carrier efficiency of cationic surfactant against cerium-bond coordinate, a new complex interaction of recently reported cerium complex was tested with synthesized cationic surfactants by conductometric measurements 22. The aim of FTIR- that there are no cation–cationic surfactant interactions in the aqueous solution of the mixed surfactants cationic and nonionic components of the cation. According to this experimentally, we contract that the surfactant Hy-1622 does not form a complex with the cerium complex and that the reduction of the free surfactant cation concentration in the solution causes a reduction of the leveling labor of the surfactant in the system involving the same charged cerium ion and cationic surfactant. The importance of vibrational frequencies of the Hy-1622 with a molecule in the C–H stretching region is shown in Figure 3. shows the FT-IR spectra before and after adding 2-DEHEHP measured for the representative sample containing 65 wt% of (Ce(NO3)2OH.3H2O. 2-DEHEHP) particles in emulsion and 35 wt% of Hy-1622 cationic surfactant together with pure Hy-1622 reference. The spectra of the control sample consist of characteristic peaks ascribed to the following vibrations at around 4000-500 cm-1 to the (CH3-CH3) frequencies are observed in 3813.78, and 3500.49 cm−1, respectively22, 23, 24. The CH3 feature located in a frequency lower than 3548.43 cm−1 is indicative of the crystalline structure. The CH3 asymmetric stretching mode presents a value 10-12 cm−1 smaller than that observed for the molecules. {(CeO2.. 2-DEHEHP)}. Furthermore, the -CH3- mode is only 2 cm−1 lower than that found in the molecules in the emulsion, in agreement. It is necessary to assign these bands. The CH3-CH3 bands of the [Ce(NO3)2 OH.3H2O. 2-DEHEHP] molecules are difficult to be observed in (b) due to their lower intensities [25,26]. It is only possible to detect them with packing densities larger molecules. It is important to indicate that it is also difficult to distinguish between CH3- and -N–CH3- bands, observing only a large band with low intensity in the region between 2952.62 and 3034.27 cm−1. O-H stretching, 2351.33-2094.63 cm-1 to the C-H stretching, 1768.30 cm-1 to the N–O bands stretching vibrations in nitroalkanes occur near 1609.34-1509.04 cm-1 carbonyl stretching vibration, 1469.16 cm-1 the C-H bending, 1288.12 cm-1 the C-O-C symmetrical stretching, 921.63 and 832.97 cm-1 the C-H bending kidding as well as 712.06 cm-1 ascribed to the buckle vibrations of Hy-1622 chains, respectively. IR spectra of the Hy-1622 in (Ce(NO3)2OH.3H2O. 2-DEHEHP) hybrid material is essentially the same with one exception in the appearance of an extra band at 562.24 cm-1 27, 28, 29. This peak is associated with the vibrations of the Ce-O bonds corresponding to intrinsic stretching vibrations of the metal cation Ce(NO3) OH.3H2O at tetrahedral in nitrate medium. This fact gives straightforward evidence of proper incorporation of the cerium nitrate particles into the Hy-1622 with emulsion.
Figure 3.FT-IR spectra (a) before adding 2-DEHEHP and (b) after adding 2-DEHEHP
Transport of Ce4+ Mechanism
The cerium ion is transported from the aqueous phase or feed into the receiving phase via the oil membrane. The movement of charge cerium ion species through the DEHEHP, B (15%v/v) as the hydrophobic liquid membrane is achieved, by the presence of a cooperative factor collected by DEHEHP, B. After complexation of the carrier with Ce(IV) on the source outline phase of the membrane, the DEHEHP, B-Ce(IV) distribute, down its concentration gradient. On the receiving phase of the membrane, the metal ions species would be released into the receiving phase across the formation of a ternary complex from carrier/Ce (IV)/sodium nitrate). At this phase, the free carrier diffuses back via the liquid membrane. The data is the transport of cerium ion from the aqueous phase into the aqueous receiving phase via the bulk of the oil membrane phase 30, 31, 32, described a schematic diagram of this mechanism is shown in equations (2&3).
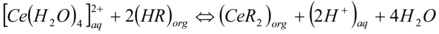 …1at pH <1.8….(2)
…1at pH <1.8….(2)
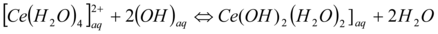 …2 at pH >2.5…(3)
…2 at pH >2.5…(3)
Where HR that the Di-(2-ethylhexyl) phosphoric acid extractant and the aq. and org. denotes aqueous and organic phase conditions respectively. At pH>2.0 the precipitate formation of Ce(OH)2 tends to diminished the efficiency, Ce(IV) aqueous concentration decreasing the Cerium extraction.
Effect of Concentrated carrier-complexation with Molar Conductances
Role of diversified constituents of organic phase during the liquid surfactant membrane formulation Feed phase at pH =2.5 has a very strong effect on the concentration of acidic dissociated (H+ + A−) as (2-ethylhexyl) phosphoric acid in H+,[NO3]-2.In a typical experiment, 15 ml of the required metal ion (5.0 ×10-4 M) was placed in the titration cell, thermostated at 250C, and the conductance of the solution was measured. Then, a known amount of a concentrated DEHEHP, B solution (1×10-3M) using a calibrated micropipette. The conductance of the solution was measured after each addition. The ligand solution (L) was added continually until the required (L: M) ratio was achieved. The formation constants, Kf, and the molar conductance (ɅM), of the resulting 1:1 complexes between 2DEHEHP, B and the Ce (IV) cation used, in different mixtures and at different from 10-250C, were calculated by fitting the observed molar conductance, Λobs, at varying DEHEHP B/[Ce4+] mole ratios to express the Λobs as a function of the free and complexes Ce(IV) ions. Figure 4 A nonlinear least-squares curve fitting using the computer software program was applied to appreciate formation, stability, and determining that molar conductance of the resulting are equally complexes 31. In the present work, all of the measured solutions were adjusted in pH range 1–2.5 using a pH-meter of the type B-417 HANNA instrument, and the prevailing cerium species were in the form of Ce(NO3)3. Figure 5. The value indicates that DEHEHP, B is an appropriate extractant to work as a phase transport catalyst to separate Ce(IV) cation and another cations transport (water/Oil/water) through DEHEHP,B in the bulk liquid membrane system.
Figure 4.Ratio between the molar conductance versus (DEHEHP,B/Ce(IV) complex in 1,2-dichloroethane.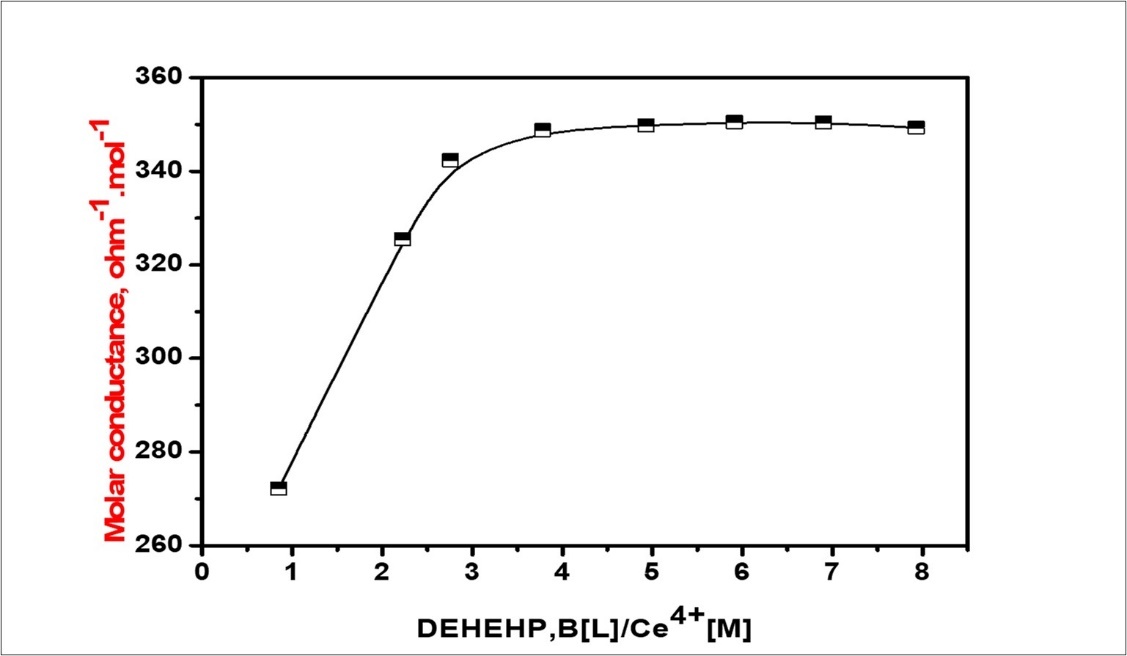
Figure 5.metal ions transport (water/Oil/water) studies for DEHEHP,B.
This may be due to the size of the Ce4+(r = 1.05 A˚) ion and, therefore, it can connect a fit condition for the DEHEHP, B (r=2.8A˚) cavity. Another data, we preliminarily investigated on competitive transmission, of cerium ion among some various metal cations and affected of surfactants also. Table 3. Study the effect of type surfactants on interfering ions source phase (5 ml) at pH=2.5. It was found that DEHEHP, B has the required ability to transport Ce(IV) cation against its concentration gradient through the bulk liquid membrane.
Table 3. The effect of nonionic and cationic surfactants on interfering ions source phase (5 ml) at pH=2.5.| Interfering ions | Surfactant | Results |
| Span 80 only | The concentration of surfactant Span 80 of Ce(IV) by analysis of different parameters showed that the nonionic surfactant does not affect K (I), Mg (II), Ca(II) while Ce(IV) slightly extraction effectiveness at pH 2.5 | |
| K (I) | 0% | |
| Mg (II) | 0% | |
| Ca (II) | 0% | |
| Ce (IV) | 34% | |
| Span 85 only | The concentration of surfactant Span 85 of Ce (IV) by analysis of different parameters showed that the nonionic surfactant does not affect K (I),Mg (II), Ca (II) 1% while Ce (IV) increasing extraction effectiveness at pH 2.5 | |
| K (I) | 0% | |
| Mg (II) | 0% | |
| Ca (II) | 1% | |
| Ce (IV) | 56% | |
| Hy-1622 only | The concentration of surfactant Hy-1622 of Ce(IV) by analysis of different parameters showed that the cationic surfactant effect on K (I), Mg (II), Ca (II) while Ce(IV) increasing extraction effectiveness at pH 2.5 | |
| K (I) | 2% | |
| Mg (II) | 10.50% | |
| Ca (II) | 25% | |
| Ce (IV) | 82% |
Extraction of Ce (IV) by LSM
The optimal conditions parameter indicate a stable emulsion is emulsification speed: 2000-4000 rpm,
emulsification time at 3 min, agitation speed: 300 rpm, internal phase concentration (NaNO3):1x10-3M carrier concentration (DEHEHP, B): 22% (w/w), Hy-1622 surfactant concentration and 3% Span 80/85 (w/w), volume, the ratio between the membrane phase and internal phase 1:3 and the volume ratio between the emulsion phase and the external phase as 1:1, 1, 2-dichloroethane as diluent. The chemical structure of the Hy-1622 surfactants was confirmed using FT-IR spectra characterization and biological activity of colloidal cerium. Besides, the cationic surfactant showed a high recurrence estimated in a period of comparative is to transport the ultimate amount of waste during the treatment process. The calculated diagram of prevalence, on a species of Ce(IV) enhances, these observations in Figure 6. The hydrolyzed species of cerium cation are probable at most coexist at pH < 3. Where aqueous, species are not the only ones affected by pH. At higher pH, the cerium hydroxide compound 33, 34 prevalent and may subsequently precipitate spontaneously without any current depending on the degree of H+ ion of an aqueous solution. This compound has been present already. The extraction of Ce(IV) higher when the feed phase concentration is in the range of HNO3 up to 7M as in Figure 7. This is prospective, because the formation of the complex between cerium ion species and extractant is most preferable, at (1-7 M) acid concentration. When the reaction rate becomes high, the driving force for mass transfer of the (DEHEHP, B-Ce(IV)) complex through the water-oil proceeds increases, and the metal complex moves on to the oil phase. Finally, the rate of the DEHEHP B complex formation depends directly on the H+ion concentration in the external phase depending on agitation speed at 300 rpm, therefore increasing the agitation speed value for the extraction phase, and the removal efficiency will be increased 35. The same removal efficiency for the studied concentration range of Ce(IV) is due to the stable amount of carrier in the experiments. The cerium ion complex should be diffused through the membrane phase to the more inner area of the globule to cast cerium ion in the internal phase.
Figure 6.prevalence diagrams of Ce(IV) species.
Figure 7.Effect of external phase concentration on removal efficiency.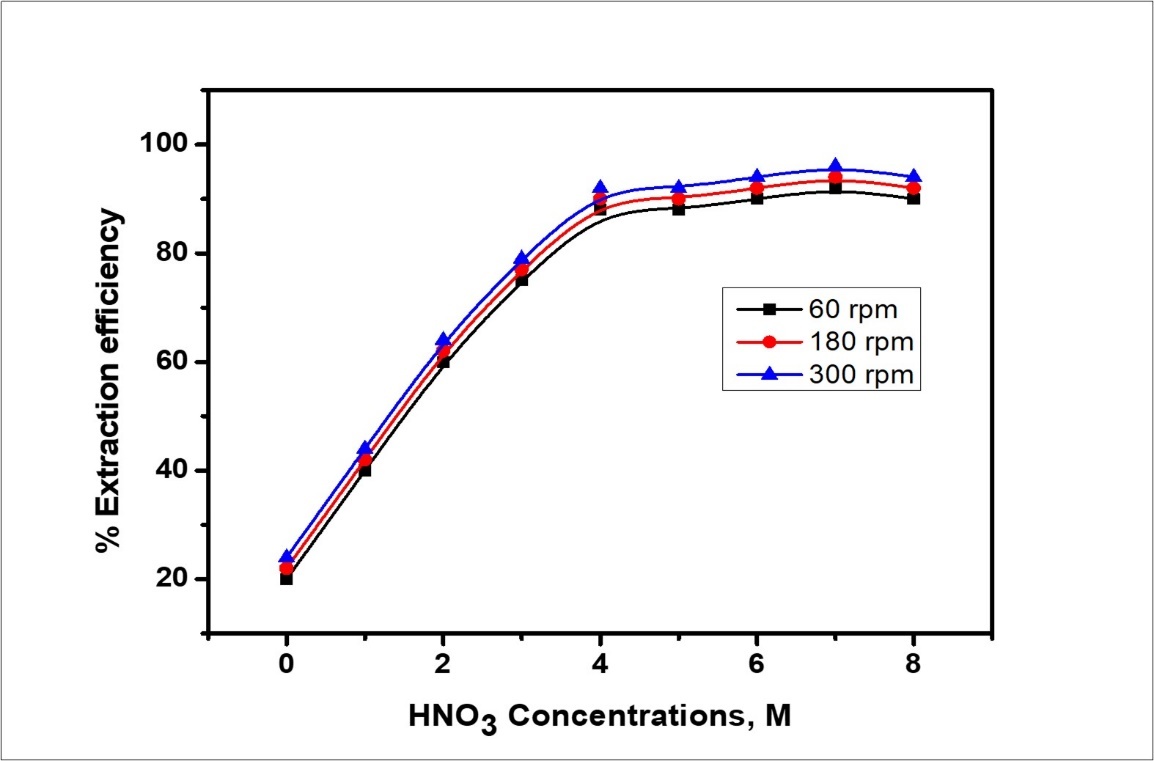
(Figure 6) shows the possibility of studying the role of ionic dissociation on the effect of surfactants in the emulsion carrier. Assuming that, in each range of Hy-1622 concentration, the cation surfactant and substrate can relate together and an equilibrium relation exists. The Hy-1622 concentration in which the equilibrium relation between the added cationic surfactant and the ions species existing in the solution, study the equilibrium between cationic-nonionic surfactants and complex produced from equilibrium surfactants and equilibrium equations (4,5). Cerium ion species in previously Figure 6 represent Ce(OH)4 or Cespecies in equilibrium step 1 and 2(e1,2) and S (A,C) anionic and cationic surfactants.
 …….(5)
…….(5)
Effect of Surfactant Concentration
Experiments were performed under the optimal conditions as mentioned previously with a carrier concentration. In a benzethonium chloride as an LSM system, a surfactant added as an emulsifier in the liquid membrane phase affects not only the stability of the liquid membrane but affect the swelling of the emulsion and the rate of cerium ion extraction. Initiatory experiments were proceeding, using benzethonium chloride (Hy-1622) and Span, 80/85 (3:1) as a cationic surfactant. Data indicated that Hy-1622 gives higher breakage values while Span 80/85 due to lower break-up of the emulsion. Thus Span 80/85(3:1) was chosen as a nonionic surfactant in this study 36. Figure 8 Explains the effect of various surfactant concentrations on emulsion stability. Although the emulsion was predictable to be stabilized with increasing surfactant concentration as a function on the degree of stability becomes approximately fixed led to the saturation of nonionic and cationic surfactants at the O/W interface. It is known that the increase in the concentration of surfactant does not countenance extraction kinetics 33. The amount of surfactant in the membrane must be lower, but it must be sufficient to stabilize the emulsion.
Figure 8.Effect of Span 80, Span 85 and Benzathonium chloride surfactant concentration on the emulsion stability.
Effect of Oily Phase Viscosity
The volume fraction of the stripping phase and the viscosity of the oily phase determine the viscosity of the W/O emulsion. The viscosity of the oily phase is proportional to the stability of emulsions. Subsequently, utilizing highly viscous oil as a membrane phase can supply more stable emulsions. However, it has been found that the increased viscosity of the membrane safely decreases the diffusivity for anisotropic fluids. Thus, this medication reduces a solute diffusivity, and consecutive lowering in the extraction rate has been specified, as a movable, in the ELM process.
Interfacial Tension of Cationic/Anionic Surfactant
Interfacial tensions in separate ions by using cationic/anionic surfactant mixtures in one aqueous phase-, without and with NaNO3 added were determined by the spinning drop method at 25oc. The relation between interfacial tension and the concentration cationic/anionic surfactant mixtures determined by the density and viscosity is shown in Table 4.
Table 4. Effect of density and viscosity data of different surfactants at different temperatures.| T=25oC | Kgm-1 ρ | (mpa)ƞ | span80ⱴ | Kgm-1 ρ | (mpa)ƞ | span85ⱴ | Kgm-1 ρ | (mpa)ƞ | Hy-1622ⱴ |
| 0 | 983.2 | 0.371 | 3.77x10-4 | 989.3 | 0.381 | 3.85x10-4 | 992.3 | 0.384 | 3.86x10-4 |
| 0.2 | 951.7 | 0.611 | 6.427x10-4 | 962.7 | 0.712 | 7.49x10-4 | 989.7 | 0.742 | 7.49x10-4 |
| 0.4 | 923.5 | 0.763 | 8.26x10-4 | 953.8 | 0.873 | 9.12x10-4 | 978.8 | 0.893 | 9.12x10-4 |
| 0.6 | 848.6 | 0.814 | 9.59x10-4 | 924.3 | 0.921 | 9.72x10-4 | 967.4 | 0.941 | 9.72x10-4 |
| 0.8 | 823.44 | 0.81 | 9.83x10-4 | 843.5 | 0.92 | 1.09x10-3 | 895.3 | 0.93 | 1.03x10-3 |
| 1 | 796.5 | 0.774 | 9.71x10-4 | 805.7 | 0.835 | 1.03x10-3 | 822.4 | 0.845 | 1.02x10-3 |
| 1.2 | 664.11 | 0.736 | 1.1x10-3 | 728.3 | 0.827 | 1.1x10-3 | 798.3 | 0.833 | 1.04x10-3 |
| 1.4 | 584.32 | 0.685 | 1.17x10-3 | 687.4 | 0.769 | 1.11x10-3 | 756.3 | 0.775 | 1.02x10-3 |
| T=35oC | Kgm-1 ρ | (mpa)ƞ | pan80ⱴ | Kgm-1 ρ | (mpa)ƞ | span85ⱴ | Kgm-1 ρ | (mpa)ƞ | Hy-1622ⱴ |
| 0 | 962.7 | 0.337 | 3.5x10-4 | 974.5 | 0.345 | 3.5x10-4 | 985.3 | 0.361 | 3.66x10-4 |
| 0.2 | 935.2 | 0.578 | 6.18x10-4 | 953.2 | 0.705 | 7.39x10-4 | 974.2 | 0.687 | 7.05x10-4 |
| 0.4 | 911.2 | 0.685 | 7.5x10-4 | 936.4 | 0.796 | 8.50x10-4 | 956.7 | 0.756 | 7.90x10-4 |
| 0.6 | 830.2 | 0.712 | 8.5x10-4 | 915.3 | 0.897 | 9.80x10-4 | 945.2 | 0.866 | 9.2x10-4 |
| 0.8 | 814.6 | 0.789 | 9.68x10-4 | 820.3 | 0.842 | 1.02x10-3 | 886.7 | 0.897 | 1.01x10-3 |
| 1 | 768.2 | 0.712 | 9.26x10-4 | 789.3 | 0.802 | 1x10-3 | 812.3 | 0.911 | 1.12x10-3 |
| 1.2 | 637.4 | 0.653 | 1.02x10-3 | 712.3 | 0.798 | 1.1x10-3 | 787.6 | 0.82 | 1.04x10-3 |
| 1.4 | 547.6 | 0.579 | 1.05x10-3 | 624.9 | 0.687 | 1.09x10-3 | 723.5 | 0.66 | 9.1x10-4 |
| T=40oC | Kgm-1 ρ | (mpa)ƞ | span80ⱴ | Kgm-1 ρ | (mpa)ƞ | span85ⱴ | Kgm-1 ρ | (mpa)ƞ | Hy-1622ⱴ |
| 0 | 956.3 | 0.321 | 3.35x10-4 | 969.3 | 0.356 | 3.67x10-4 | 978.3 | 0.375 | 3.8x10-4 |
| 0.2 | 947.6 | 0.6 | 6.33x10-4 | 943.7 | 0.7 | 7.41x10-4 | 962.6 | 0.712 | 7.39x10-4 |
| 0.4 | 914.5 | 0.723 | 7.9x10-4 | 936.8 | 0.842 | 8.9x10-4 | 954.3 | 0.865 | 9.06x10-4 |
| 0.6 | 814.6 | 0.802 | 9.9x10-4 | 919.3 | 0.902 | 9.81x10-4 | 936.2 | 0.922 | 9.84x10-4 |
| 0.8 | 818.4 | 0.809 | 1.01x10-3 | 823.7 | 0.911 | 1.10x10-3 | 874 | 0.928 | 1.06x10-3 |
| 4870451141730001 | 754.5 | 0.764 | 1.12x10-3 | 798.7 | 0.815 | 1.12x10-3 | 811.7 | 0.836 | 1.02x10-3 |
| 1.2 | 632.3 | 0.712 | 1.1x10-3 | 718.6 | 0.808 | 1.1x10-3 | 769.2 | 0.82 | 1.06x10-3 |
| 1.4 | 567.12 | 0.549 | 9.68x10-4 | 656.4 | 0.685 | 1.04x10-3 | 736.3 | 0.747 | 1.01x10-3 |
| T=45oC | Kgm-1 ρ | (mpa)ƞ | span80ⱴ | Kgm-1 ρ | (mpa)ƞ | span85ⱴ | Kgm-1 ρ | (mpa)ƞ | Hy-1622ⱴ |
| 0 | 944.8 | 0.318 | 3.36x10-4 | 960.5 | 0.325 | 3.38x10-4 | 964.3 | 0.344 | 3.56x10-4 |
| 0.2 | 921.1 | 0.525 | 5.69x10-4 | 933.2 | 0.698 | 7.47x10-4 | 945.2 | 0.654 | 6.92x10-4 |
| 0.4 | 908.8 | 0.647 | 7.21x10-4 | 906.4 | 0.596 | 6.5x10-4 | 935.6 | 0.712 | 7.6x10-4 |
| 0.6 | 824.3 | 0.7 | 8.49x10-4 | 875.3 | 0.799 | 9.1x10-4 | 923.5 | 0.832 | 9.01x10-4 |
| 0.8 | 808.9 | 0.772 | 9.54x10-4 | 811.3 | 0.833 | 1.02x10-3 | 838.7 | 0.847 | 1.0x10-3 |
| 1 | 742.6 | 0.674 | 9.07x10-4 | 765.3 | 0.759 | 9.91x10-4 | 800.3 | 0.902 | 1.12x10-3 |
| 1.2 | 624.8 | 0.622 | 9.96x10-4 | 702.3 | 0.778 | 1.10x10-3 | 747.5 | 0.803 | 1.0x10-3 |
| 1.4 | 523.8 | 0.509 | 9.7x10-4 | 612.3 | 0.668 | 1.09x10-3 | 698.5 | 0.647 | 9.2x10-4 |
Effect of Treat Ratio of Vm Into VCe(IV)
The treat ratio, defined as the volume ratio of the emulsion phase (Vm) to the aqueous external solution (VCe), an important key in determining the efficiency of ELM. Through experiments were to study the effect of treat ratio on the extraction of cerium ion species. It's not changeable random, because it is necessary to pledge that the HNO3 concentration that's existent in the internal phase is high enough to react with the external phase which is the cerium nitrate solution. The flow rate ratio of the dispersed phase to the continuous phase was varied from (Vm: VCe =1:2) to (Vm:VCe = 1:50) by confirmation, the flow rate of the dispersed phase as 25 ml/min and changing the flow rate of the continuous phase, which is the external phase, from 25 to 100 ml/min. The influence of the treat ratio of Vm into VCe(IV) is shown in Figure 9. It is found that at a treat ratio of (1:2), the achievement, recovery of solute in the external phase is high, and an increase in treat ratio reduces the removal efficiency. It is apparent that the volume ratio between the feed into emulsion solution decrease due to the removal efficiency also decreases. Additionally, the effect of (Vem) the volume ratio of emulsion to the external feed (Vfeed) solution on the stability is found to be derivative, because the breakage increases very slightly with an increase of this volume ratio. Also, the increase in the treat ratio with a reduced external volume phase, the volume of emulsion will be increased. Thence, increasing the surface area for mass transfer due to the formation of a larger number of emulsion globules. In this work that emulsion type is dependent on the relative phase volume concluded that at a phase volume to the emulsion would be congested, more densely than was possible. This means that any attempt to exceed a phase volume of ɛ= 1.07 for the internal phase has to be data in each, inversion, or breaking. Figure 10 declares the effect of the volume ratio of oil to water O/W on emulsion stability at the Hy-1622 dosage of 1:2. When the ratio of O/W was decreased gradually, from 1:10 to 1:1, the comparative volume of the emulsion was increased consistently. As shown in Figure 11 after 18 h, the comparative volume of the emulsion was 16.27% in the ratio of 1:10. The relative volume of the emulsion was 27%, 47%, and 85%, at 12 h,18hr, and 20 h, respectively 37, 38, 39. When the ratio was 1:1 at ½ h, the relative volume was 97.3%. At this ratio, the relative volume was ultimately constant throughout 20 h, indicating that the emulsion the best stability.
Figure 9.Effect of Benzathonium chloride dosage on emulsification.
Figure 10.Effect of O/W ratio on emulsion stability at the Hy-1622 dosage of 1:2.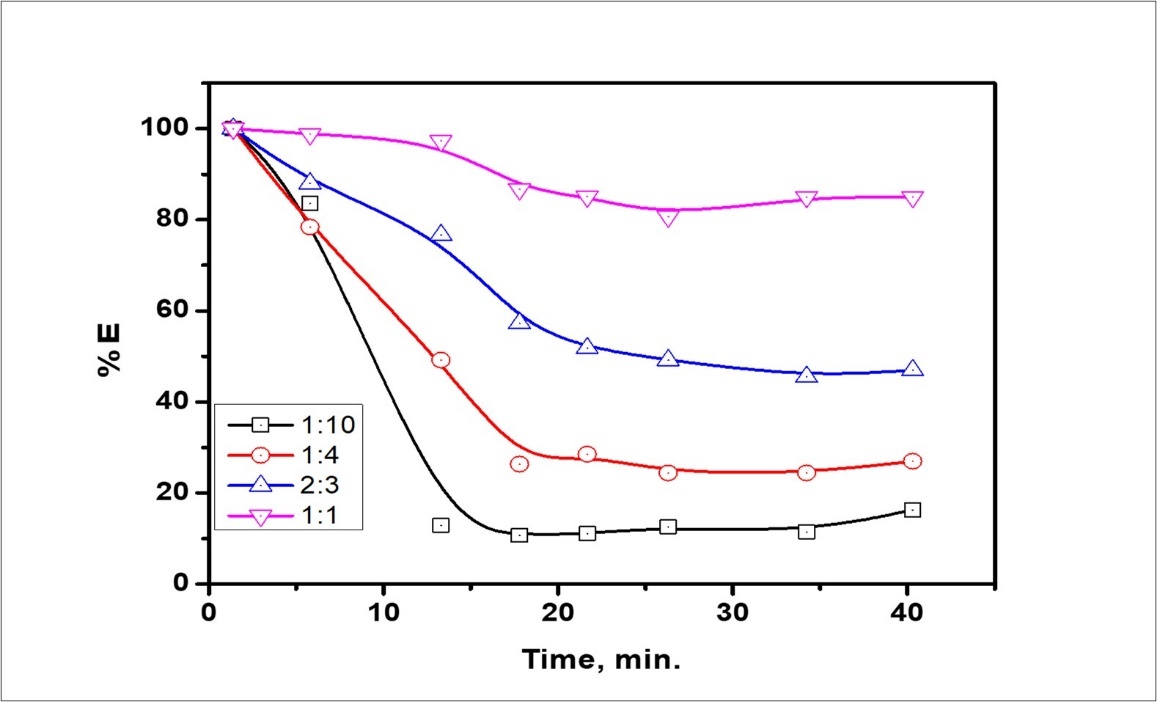
Figure 11.Effect of stirring speed on benzathonium chloride emulsification at O/W 1:1.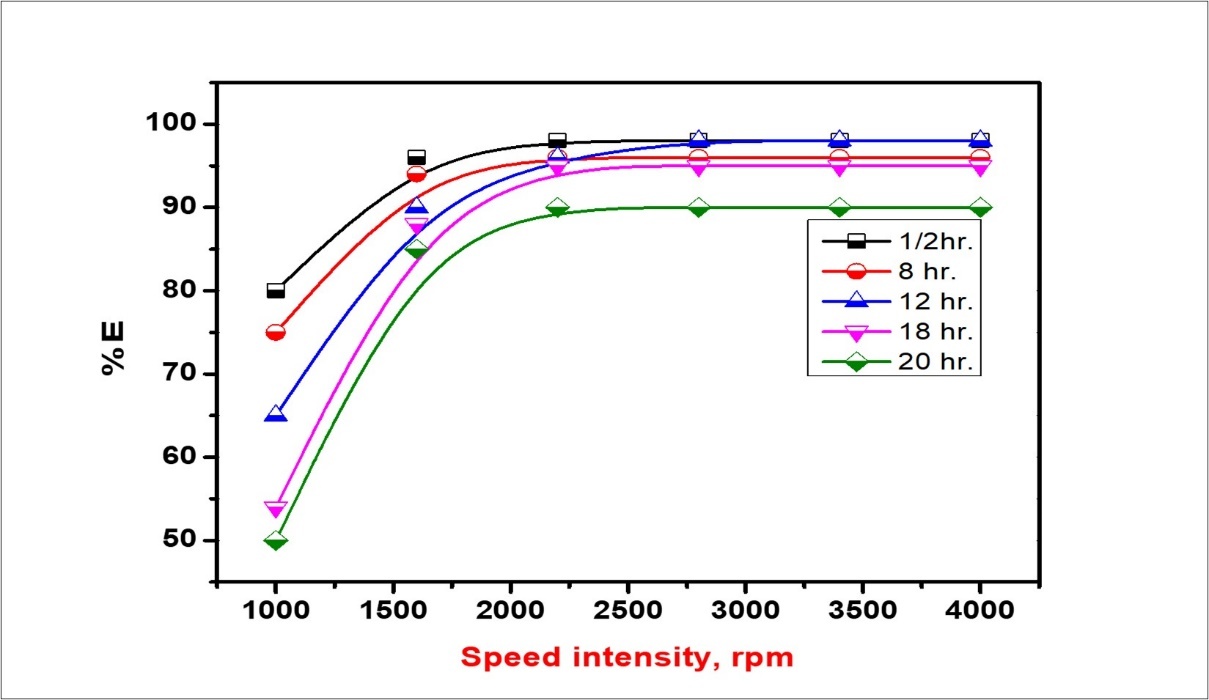
Effect of Stirring Rate
To prepare emulsions of two liquids that are insoluble or dominate only slight reciprocal, solubility. Benzethonium chloride as emulsification is usually accomplished, by the application of mechanical energy. At first, the interface between the two liquid phases is distorted to such a dimension, that large droplets are established, and these large droplets are posteriorly separate, into a minimum. During emulsification, the interfacial area between two aqueous solutions increases. Liquids lead to minimizing this surface area; thus, the driving force is required for emulsification to proceed. This work described that increasing domestic, separation, of driving force in the breaking area, lead to the high circulation exhaustion through the mixer area is found to be the best effect. Method of drops reduced diameter. The purpose of stirring is to format, of stable and emulsion of homogeneous by breaking large liquid drops into smaller drops. Figure 12 illustrates the proportional, volume of the emulsion as a function of stirring speed after stirring the mixture for 20 hr. The relatively, the volume of emulsion which prepared at least 1000 rpm stirring rate will decrease, sharply with time, elucidate, that a 1000 rpm was not adequate for this emulsion technique. The best display, the reliance, of emulsion stability on stirring rate, the relative emulsion volume after 20 hr, stirring followed by 10-h remain is plotted against time in Figure 12 which obviously, indicates that the stability of emulsion was accomplished with higher stirring intensity. A stirring speed higher than 3850 rpm was, however, not desired because the higher stirring intensity will drive the emulsifier to break away from the oil-water interface.
Figure 12.The relationship between stirring speed and % E volume after 20 h.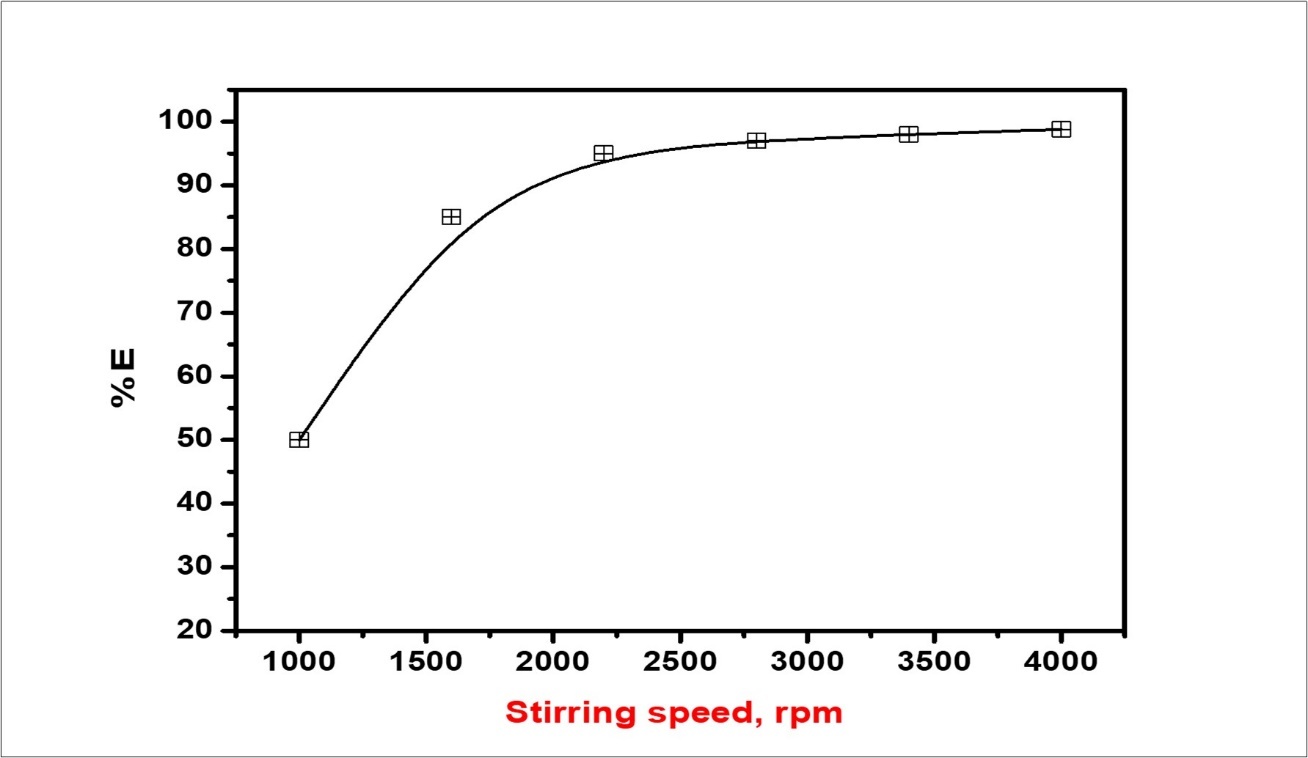
Effect of Temperature
The transfer of cerium ion species into the emulsion is facilitated due to reducing in viscosity by rising temperatures. Then, the stability of the emulsion is reduced at increasing temperatures. Figure 13 compares the removal efficiency at five different temperatures of 25, 35, 45, 65, and 75◦C. Figure 13 shows that the maximum extraction qualification, are obtained at lower temperatures. Then, the maximum extraction efficiency at temperature is lower than that at 25◦C that indicates the surrounding temperature is optimal for this process.
Figure 13.Effect of mixing temperature on cerium ion extraction efficiency using Benzathonium chloride surfactant.
Effect of Mixing Time
The effect of mixing time MT on the emulsion stability was tested. Experiments under optimal conditions used were corresponding, to those used formerly. The MT was different, from 0 to 35 min. Figure 14 displays the effect of MT on the stability of the emulsion. The data indicate that the (%E) of the emulsion increases with increasing the mixing time, emulsion stability decreased. This experiment also has seen that the mean size (d32) of the particles decreased very rapidly in the first few seconds and then progressively, acquire, the limiting value after 18 min. To best seen the subjection of emulsion stability on mixing time, the comparative emulsion volume after one day is versus against mixing time in Figure 14 which distinctly indicates that the optimal mixing time is 18 min.
Figure 14.The relation between mixing time and % extraction efficiency of emulsion after one day.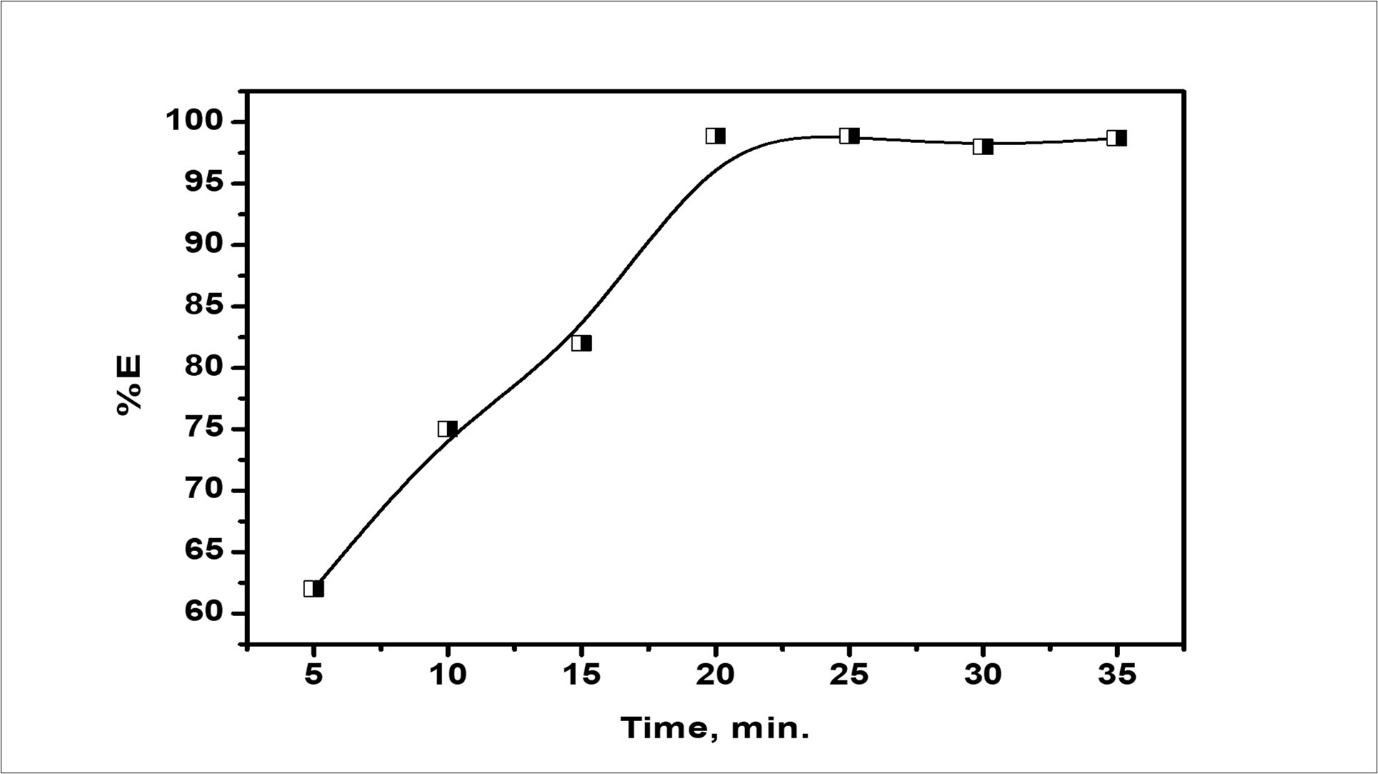
Conclusion
The removal of cerium ions (IV) species from aqueous nitrate solution was studied by ELM technique, in the presence of C64H124O26 with C60H108O8 as a non-ionic surfactant synergistic with the cationic benzethonium chloride, which plays a major role in stabilizing the emulsion. The present study showed that emulsions made with Span 80 / Span 85 (3: 1) in the absence of other than Hy-1622 chloride are very stable and show low extraction efficiency. The extraction efficiency increases with increasing the dose of Hy-1622 up to a certain concentration of surfactant and then decreasing due to the dominating effect of the surfactant on the mass rate transfer. The stability of the emulsion is increased by reducing the ratio of the emulsion O / W to the small oil droplets dispersed in the aqueous phase. The preferable, ratio was 1:1 by volume. Higher stirring speed is produced in a more stable emulsion. The optimal stirring speed was 4000 rpm for the emulsion system investigated. High temperatures are reduced, and the best emulsifying temperature was 250C according to the density and viscosity data of different surfactants at different temperatures. The stability of emulsion increased with stirring speed up to 18 min, behind which the stability, reduced with furthermore higher in stirring time led to the withdrawal of surfactant from the oil-water interface. The emulsion liquid membranes technique for extracting and recovering Ce (IV) in the presence of Hy-1622 as a cationic surfactant has the advantages of low cost, high efficiency, environmental protection, and increased potential in industries.
References
- 5.Rivoira V A Valles L P, M L, Sa-ngasaeng Y, Jongpatiwut S, A R Beltramone et al.. Today (2019), https://doi.org/10.1016/j.cattod.2019.08.005
- 7.SM Hunagund Pujar, Barretto D A, Desai V R, Patil S, Vootla S K et al. (2019) . , Mater. Sci 43, 1-11.
- 13.LMS Hussin Mohamed, Gad HMH, Daifullah AAM, SA. (2013) . , Abo-El-Enein, J. Memb. & Sep. Tech 2, 102.
- 15.T C FERN. (2012) Thesis submitted in partial fulfillment of the requirements for the award of the degree of Bachelor of Chemical Engineering (Gas Technology).
- 22.Tikhomirov M, Pierunek N, Śniadecki Z, Idzikowski B, Marędziak M et al. (2018) . , J. Mater. Scie. & Enge 90-248.
- 23.Suresh S, Karthikeyan S, Jayamoorthy K. (2016) . , J. of Sci. Advanced Materials and Devices 1, 343.
- 24.Saravanan P, Jayamoorthy K, S A Kumar. (2016) . , J. of Sci. Advanced Materials and Devices 1(3), 367.
- 25.Pązik R, Zięcina A, Zachanowicz E, Małecka M, Poźniak B et al. (2015) . , Eur., J. Inorg. Chem 4750.
- 26.Suresh S, Karthikeyan S, Saravanan P, K Jayamoorthy. (2016) . , Intern. J. of Modern Science 2(3), 188.
- 29.Suresh S, Karthikeyan S, Saravanan P, Jayamoorthy K, I K. (2016) Dhanalekshmi Karbala Intern. , J. of Modern Science 2(2), 129.
- 33.C M Browner, Frace S, Robert P, M B Rubin. Development Document for Best Available Technology, Pretreatment Technology, and New (1994) Source Performance Technology for the Pesticide Formulating, Packaging, and Repackaging Industry 821.
- 35.Fernández-Bolaños J, Rodríguez G, Rodríguez R, Guillén R, A. (2006) . , Jiménez GRASAS Y ACEITES 57(1), 95.
Cited by (1)
- 1.Nseke Joseph Makela, Kabuba John, Otunniyi Iyiola Olatunji, 2025, Adsorption of selected oxyanions from aqueous solution using Benzethonium chloride modified zeolite 4A: Artificial neural network approach, Microporous and Mesoporous Materials, 382(), 113395, 10.1016/j.micromeso.2024.113395

